


IVF Embryo Genetic Testing
Preimplantation Genetic Testing may increase the probability of success


Preimplantation Genetic Testing (PGT) of embryos has been recommended for use alongside In Vitro Fertilization to identify and transfer only genetically normal embryos, aiming to increase the chances of a successful pregnancy.

IVF Embryo Genetic Testing
Preimplantation Genetic Testing (PGT), in conjunction with In Vitro Fertilization (IVF), aims to increase the likelihood of a successful pregnancy by identifying and transferring genetically normal embryos while decreasing the chances of transferring genetically abnormal embryos.
Embryo genetic testing has been suggested for patients who are at risk of an elevated probability of having genetically abnormal embryos. This includes women of advanced maternal age (35 or older).
Additionally, it may provide benefits for patients with a history of repeated early pregnancy loss (three or more), repeated failed IVF cycles despite transferring high-quality embryos (three or more transfers), and severe male factor infertility.
Preimplantation Genetic Testing (PGT) is a multifaceted treatment that involves several steps. It includes the creation of embryos through IVF, micro-biopsy of normally developing embryos, embryo cryopreservation, genetic analysis of the extracted cells (PGT), and finally, a Frozen Embryo Transfer.

IVF with PGT Treatment Process

1 — IVF to create embryos
In Vitro Fertilization procedure creates as many high-quality embryos as possible

2 — Extended embryo culture to blastocyst
By the fifth to seventh day after the egg retrieval, the embryos should reach the blastocyst stage (80 or more cells)

4 — Embryo Freezing
Since it takes several days to carry out the embryo genetic analysis, the blastocysts are cryopreserved immediately after their biopsy and stored in liquid nitrogen in our IVF laboratory

3 — Blastocyst trophectoderm biopsy
An embryo biopsy is performed by creating an opening in the eggshell around the embryo. It is possible to safely remove six to eight cells through this opening using a special microscope with micromanipulators

5 — Genetic analysis of the embryonic tissue
The genetic analysis will check for many of the most common genetic conditions and will also determine the gender of each embryo
6 —Liquid nitrogen storage of the embryos
Frozen embryos can be stored for extended periods of time, but most patients will start their Frozen Embryo Transfer cycle with the onset of the first menstrual period after their IVF treatment

7 — Subsequent Frozen Embryo Transfer
When you decide to conceive with your cryopreserved embryos, one or two genetically “healthy” embryos of the desired gender will be thawed and transferred into your uterus

IVF Embryo Genetic Testing Procedure
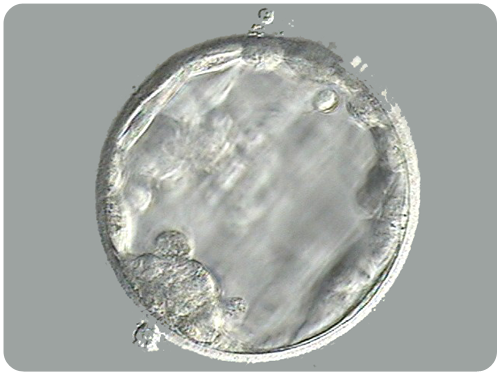
IVF blastocyst culture
Embryos should reach the blastocyst stage (80 or more cells) by the fifth to seventh day after the egg retrieval.
The picture below shows an advanced stage of blastocyst development. Notice the central fluid-filled cavity.
The cells within the blastocyst have already differentiated into the inner cell mass (at seven o’clock) that will give rise to the fetus and the trophectoderm cells that will form the future placenta.

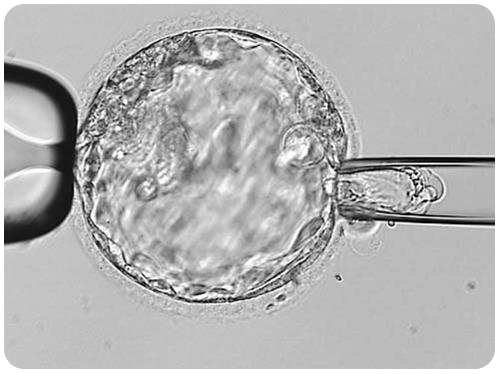
Blastocyst trophectoderm biopsy
An embryo biopsy is performed by creating an opening in the eggshell around the embryo. It is possible to safely remove six to eight cells through this opening using a special microscope with micromanipulators.
The cells are taken from the trophectoderm only (future placenta cells, genetically identical to embryonic cells). The “inner mass” cells (embryonic cells) are not removed.
So far, there is no evidence that embryonic biopsy results in an increased chance of abnormalities in the baby or that the risk of birth defects is higher when compared to conceptions that occur spontaneously without medical assistance (2% to 5%)


Blastocyst cryopreservation
Since the embryo genetic analysis takes several days, the blastocysts are cryopreserved immediately after their biopsy and stored in liquid nitrogen in our IVF laboratory.

Genetic analysis of the embryonic tissue
The accuracy of PGT cell analysis of genetically abnormal embryos approaches 100% but is not guaranteed. Even though highly unlikely, it is possible that an embryo tested as normal may not be genetically perfect.


Liquid nitrogen storage
Vitrified embryos can be stored for extended periods of time, but most patients will start an embryo transfer cycle with the onset of the first menstrual period after their IVF treatment.


Subsequent Frozen Embryo Transfer
The endometrial lining is first stimulated with estrogen and progesterone, followed by the thawing of one or two embryos tested as normal by the PGT analysis and then transferring them inside the uterus.
If you are successful, your pregnancy becomes indistinguishable from conception through intercourse, and your obstetrical care should be no different than if you conceived without any treatment.

Bay IVF Early Pregnancy Heartbeat



Prerequisites for IVF with PGT
Only a few treatment prerequisites are needed before the start of your treatment. They assess the egg and sperm quality and ensure that embryos can be accurately placed in a healthy uterus.
Female partner’s age
The probability of a successful IVF treatment becomes exceedingly low by age 44. You must be 43 and 11 months or younger at the time your IVF treatment begins. Patients who are 44 and older should strongly consider Donor Egg IVF as the most effective treatment option to achieve a successful pregnancy.
Female partner’s weight
Your BMI must be no greater than 31 for an adequate ovarian response and safety during the egg retrieval procedure. If your BMI is higher, please follow the IVF Diet and Lifestyle (PDF) recommendations as closely as possible. By doing so, you can achieve weight loss at an optimal rate, which could significantly improve your chances of a successful pregnancy.
Pathogen testing
This testing is required by the State of California. You and your partner (if applicable) must be tested for Hepatitis B-Surface Antigen, Hepatitis C-Antibody, HIV I&II, HTLV I&II (male partner only), and RPR.
Ovarian Reserve Assay (ORA)
ORA assesses the likelihood of producing normal-quality eggs. It involves measuring Follicle Stimulating Hormone (FSH), estradiol (estrogen, E2), and Anti-Mullerian Hormone (AMH) blood levels.
These test results are used to optimize ovarian stimulation.
Antral Follicle Count
The number of antral follicles (small fluid-filled sacs within the ovaries seen on ultrasound) in unstimulated ovaries is related to the quality of the eggs. Ideally, there should be approximately 20 antral follicles in both ovaries combined.
Uterine measurement and saline ultrasound
It is important to determine the length of the uterus in order to ensure the correct placement of embryos.
A saline ultrasound is utilized to rule out the presence of intrauterine polyps or fibroids.
Semen evaluation
The male partner’s semen will be evaluated at Bay IVF to determine the best laboratory method for semen preparation for egg insemination.
Parents’ genetic testing (optional)
All prospective parents should consider genetic screening for hundreds of the most common genetic diseases. Please inform us during your initial appointment at Bay IVF if you would like to have your blood sample sent for genetic screening.
IVF diet and lifestyle
Environmental factors have a significant impact on reproductive health. Please review the IVF Diet and Lifestyle document (PDF) for a comprehensive list of environmental reproductive health recommendations and a source for dietary supplements.



PGT remains a personal choice for couples, particularly those without a history of repeated pregnancy losses.


FAQ — Find Answers to Your Queries
Here are some of the most commonly asked fertility-related questions. A more comprehensive FAQ selection is available on the FAQ page.
Should we transfer one or two embryos?
About half of our patients transfer two embryos at a time, while the other half choose to transfer only one embryo in each transfer. We will always provide guidance, but you will have the final say in whether to transfer one or two embryos.
What is gender selection treatment?
Gender selection is an extension of IVF treatment that utilizes the Pre-implantation Genetic Testing (PGT) procedure to identify the gender of each embryo. With this method, prospective parents can know the gender of each embryo with 100% accuracy prior to transfer into the uterus.
I am from the LGBTQ+ community. Can I have my treatment at Bay IVF?
Yes! We take pride in being one of the first fertility clinics in Northern California to offer In Vitro Fertilization services for lesbian partners. Our clinic is LGBTQ+ friendly and experienced in both lesbian IVF and Reciprocal IVF.
Is the egg retrieval procedure painful?
Your egg retrieval should be a very comfortable experience, as we use effective pain and relaxation medications. Most patients will sleep through the 5-10 minute egg retrieval procedure, though some may experience mild menstrual-like cramping.
What is the IVF cut-off age?
The probability of a successful IVF treatment becomes exceedingly low by age 44. You must be 43 and 11 months or younger at the time your IVF treatment begins. Patients who are 44 and older should strongly consider Donor Egg IVF as the most effective treatment option to achieve a successful pregnancy.
I am over 44, but my ovarian reserve is very good. Can I do IVF treatment?
We have occasionally provided IVF treatment to women over the age of 44 when their antral follicle count and AMH results suggest that their eggs are biologically younger than their chronological age.
What are the BMI requirements?
Your BMI must be no greater than 31 for an adequate ovarian response and safety during the egg retrieval procedure. If your BMI is higher, please follow the IVF Diet and Lifestyle (PDF) recommendations as closely as possible. Doing so can help you achieve weight loss at an optimal rate, which could significantly improve your chances of a successful pregnancy.
Do you offer payment plans?
Our IVF Duo and IVF Trio Financial Plans can provide substantial savings compared with single-cycle IVF fees. Two- and three-cycle financial plans for Egg Freezing are also available.
Bay IVF has partnered with Future Family to offer accessible, personal, and affordable fertility care financing. Their mission is to make fertility care more accessible and affordable. Future Family loans are an alternative to high-interest credit cards and dipping into your savings. They offer 0% APR financing for those who are eligible, as well as low monthly payment plans. Prequalifying is risk-free, easy, and will not affect your credit score.
Meet Your Doctor

- Dr. Polansky received his medical diploma from Charles University in Prague, the Czech Republic, in 1978.
- After completing his OB/GYN residency at Jewish Hospital in Saint Louis, MO, he graduated from the Reproductive Endocrinology and Infertility (REI) fellowship at Stanford University in 1985.
- In the same year, he co-founded the Stanford IVF Clinic.
- Dr. Polansky obtained board certification in Obstetrics and Gynecology in 1986 and became REI subspecialty board certified in 1988.
- In 1987, he left Stanford University and established Nova IVF.
- In 2011, he founded Bay IVF, where he provides advanced fertility treatments with a holistic approach, utilizing state-of-the-art techniques.
- Dr. Polansky personally performs ultrasound examinations, egg retrievals, embryo transfers, and ovarian and endometrial stimulations for his patients.
- He is deeply committed to his patients and is always ready to lend a helping hand.
Frank Polansky, M.D.



Initial Appointment Questions
When you call to schedule your consultation, one of our Front Office Coordinators will ask you a short series of questions regarding your reproductive history.
Your Initial Visit at Bay IVF
Attending a new patient appointment at a fertility clinic can be stressful. Our primary objective is to ensure that your initial visit is friendly and relaxing. We encourage you to ask questions at every step of the process.

1 — When You Arrive
You will be welcomed by one of the clinic receptionists. One of our nurses will measure your height and weight and take your blood pressure

2 — Meet Your Doctor
Dr. Polansky will ask you a series of clarifying questions and then provide you with a summary of the factors contributing to your infertility

4 — Exam Room
One of the nurses will escort you to an examination room. Your examination will begin with listening to your lungs and heart

3 — Ask Your Questions
You will then have a discussion with him about the most suitable reproductive treatment(s) for you. During this time, you will have the opportunity to ask any questions you may have

5 — Ultrasound of the Ovaries
The next step is a pelvic ultrasound to examine the uterus and ovaries. This ultrasound will help determine the number of antral follicles present within the ovaries

6 — Financial Part
Following that, you will have a discussion with one of the financial advisors regarding the financial aspects of your treatment, including potential treatment financing options

8 — Support 24/7
If you have any questions after leaving the clinic, please feel free to reach out to us via phone call, text, or email. Open and discreet communication is an integral part of the care we provide at Bay IVF

7 — What About Time?
Your entire visit is expected to last approximately one hour


Schedule Your Initial Consultation With Dr. Polansky
Online (free) or In-Person
Call or Text Us: 650-322-0500
You can also complete the form below to request your initial consultation


We look forward to meeting you at Bay IVF and, when your treatment is successful, celebrating your new pregnancy!








